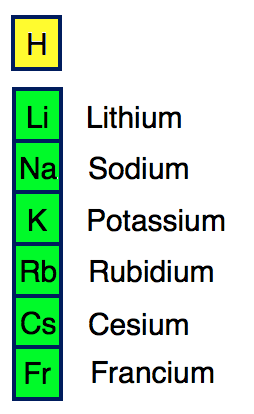 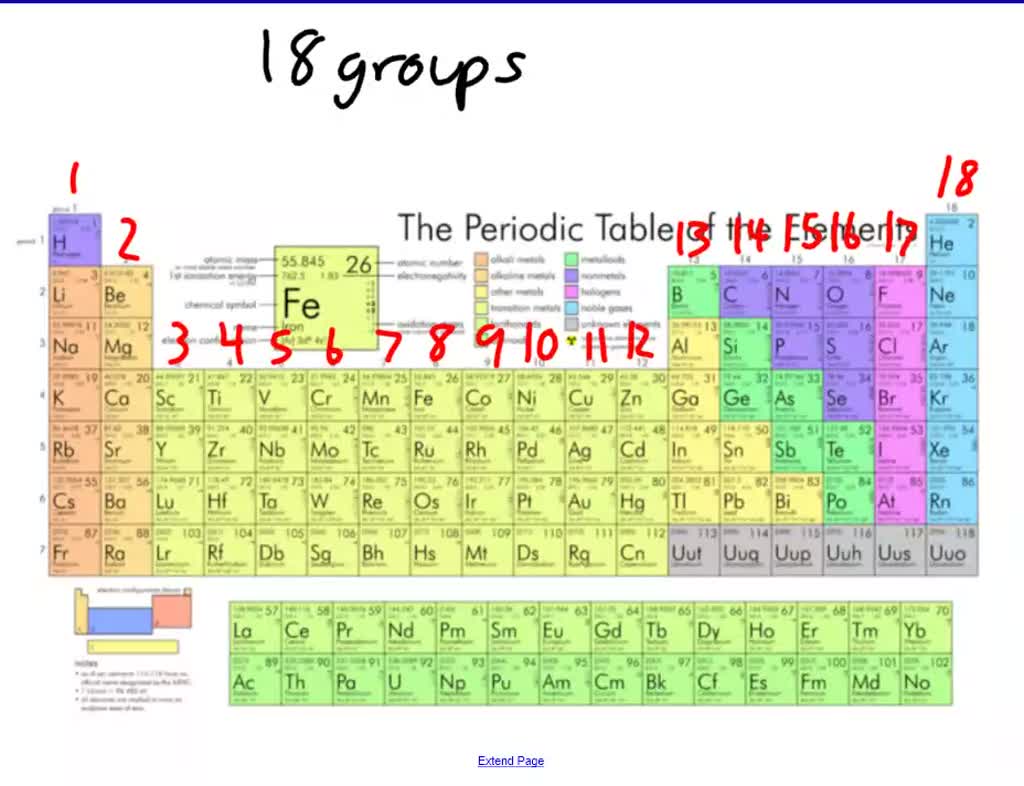
Groups of nonmetals include the nonmetals, halogens, and noble gases. The free metal does not occur in nature and must be prepared from compounds. Sodium is an alkali metal, being in group 1 of the periodic table. It is a soft, silvery-white, highly reactive metal. The alkali metals are the elements in Group 1A on the periodic table (all the elements in the first column excluding hydrogen H): lithium (Li), sodium (Na). Groups of metals include the alkali metals, alkaline earth metals, transition metals, basic metals, lanthanides, and actinides. Sodium is a chemical element it has symbol Na and atomic number 11. Elements may be further subdivided into groups. The rest of Group 1 elements tend to form comppunds in the form of MO 2 where M is K, Rb, Cs, or Fr. The common oxidation states for arsenic, antimony. While all of the metals except mercury are solid under ordinary conditions, nonmetals may be solids, liquids, or gases at room temperature and pressure. Arsenic, antimony and bismuth react with oxygen when burned. In contrast, most nonmetals are poor conductors of heat and electricity, tend to be brittle solids, and can assume any of a number of physical forms. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). Metals are generally good conductors of heat and electricity, are malleable and ductile, and have a lustrous metallic appearance. Group 4 is the second group of transition metals in the periodic table. formation to the position of the element on the periodic table, and the valence. These elements are found along a zig-zag line that runs from the upper left of group 13 to the bottom right of group 16. Respectively for the elements of Li and Na, Li is positioned in the 2nd period and has its valence electrons in the 2nd energy levels whereas Na belongs to the. (b) Sodium is in Group 1 of the periodic table with an electron arrangement. Elements that have some properties of metals and some properties of nonmetals are called metalloids or semimetals. The far right side contains the nonmetals, plus hydrogen displays nonmetal characteristics under ordinary conditions. The noble gases were often also called the inert gases, however, since noble gas compounds are now known this name is no longer used. Noble gas is translated from the German noun Edelgas, first used in 1898 by Hugo Erdmann (1862 - 1910) to indicate their extremely low level of reactivity. The elements with the most metallic character are on the left side of the periodic table (except hydrogen). The Group 18 elements have a particular name Noble gases. Metallic character is a periodic table trend. These properties include metallic luster, formation of cations, high electrical and thermal conductivity, and malleability. As a result, elements in the same group often display similar properties and reactivity. Metallic character is the set of properties associated with metals. It is in the subgroup of alkaline metals NaO sodium oxide and NaOH sodium. The elements in each group have the same number of valence electrons. Sodium is an element in the 1 group of the 3 period of the Periodic Table. They are found on the lefthand side of the table. The s-, p-, and d-block elements of the periodic table are arranged into 18 numbered columns, or groups. Indeed, the integer −1 cannot be expressed as a finite sum of 1s.Another way to categorize elements is according to whether they behave as metals or nonmetals. In other words, if S is not a monoid generator of the set of integers. In abstract algebra, a generating set of a group is a subset of the group set such that every element of the group can be expressed as a combination (under the group operation) of finitely many elements of the subset and their inverses. Sodium, symbol Na, has a Body Centered Cubic structure and Silver color. Each non-identity element generates the group. Element 11 of Periodic table is Sodium with atomic number 11, atomic weight 22.98977. If gN/p 1 g N / p 1 for some p p, then replace N N by N/p N / p and start again - note that there will be a maximum of log n log n reductions. For all primes p p dividing N N, compute gN/p g N / p. Abstract algebra concept The 5th roots of unity in the complex plane form a group under multiplication. In other words, you know a (possibly very large) integer N N such that the order of the element g g divides N N.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
